Schematic arrangement of XRD Diffractometer componentsX-ray studies are mainly carried out in two basic configurations, namely, Single crystal and Powder XRD. However, the component parts of the x-ray spectrometer are in general common and comprise of:. Source of x-rays. Sample stage.
Below is a schematic diagram for a powder X-ray diffractometer, showing the rotating detector. The source shown is an X-ray tube, which is the most common source of X-rays. Filters are used to provide a narrow wavelength range for analysis. Why is a monochromatic source desirable for XRD analysis?
DetectorThe article will provide basic details on the component parts of the x-ray diffractometer.Source of x-rays Schematic diagram of X-ray tubeX-ray tube is a common source of x-rays. It comprises of an evacuated tube which contains a copper block anode bearing a metal target made of any of the metals such as molybdenum, tungsten, copper, rhodium, silver or cobalt.The cathode is a tungsten filament.On passage of electric current through the filament electrons are generated which move towards the anode under the highly accelerated voltage typically 30 – 150 kV. The accelerating electrons on striking the metal surface knock out electrons from the inner shells and the vacancies created are filled by electrons from the outer shells. In the process metal atoms emit x-rays. However, this involves heating of the metal block and x-rays constitute only a small fraction of the total energy liberated. The emitted x-rays exit the tube through a berylium window. The copper block needs to be cooled with a supply of water to dissipate the excessive heat generated.
The Be window helps transmit a monochromatic beam of x-rays. Further monochromatization can be achieved by making use of a zirconium filter when using molybdenum as metal target. It absorbs the unwanted emissions while allowing the desired wavelengths to transmit.Sample stageSample stage is also known as sample holder or a goniometer. Single crystal diffractometers make use of 4 circle goniometers. These circles help position the crystal planes for optimum settings. The sample stage can be a simple needle that holds the crystal in place or glass plate or fiber on which the crystal is mounted using an epoxy resin.
Only sufficient quantity of epoxy resin is used so that the crystal is clearly mounted and not embedded in the resin. The fiber is mounted on a brass mounting pin and then inserted into the goniometer head. The sample is then centred with an optical arrangement such as a microscope or video camera and making adjustments along X, Y and Z directions to achieve optimum centering under the crosshairs of the viewer.DetectorsIn earlier days photographic films were used for recording the absorption pattern of diffracted beams. With the advances in detection technology more sensitive detector options were incorporated in advanced instruments.
Such detectors include gas filled transducers, scintillation counters and semiconductor transducers. Solid state detectors offer highest levels of sensitivity and speed of analysis.Subsequent articles will cover the operating principles and benefits of single crystal and powder systems.
Max von Laue, in 1912, discovered that crystalline substances act as three-dimensional diffraction gratings for X-ray wavelengths similar to the spacing of planes in a crystal lattice. X-ray diffraction is now a common technique for the study of crystal structures and atomic spacing.X-ray diffraction is based on constructive interference of monochromatic X-rays and a crystalline sample. These X-rays are generated by a cathode ray tube, filtered to produce monochromatic radiation, collimated to concentrate, and directed toward the sample. The interaction of the incident rays with the sample produces constructive interference (and a diffracted ray) when conditions satisfy ( n λ=2 d sin θ). This law relates the wavelength of electromagnetic radiation to the diffraction angle and the lattice spacing in a crystalline sample.
These diffracted X-rays are then detected, processed and counted. By scanning the sample through a range of 2 θangles, all possible diffraction directions of the lattice should be attained due to the random orientation of the powdered material. Conversion of the diffraction peaks to d-spacings allows identification of the mineral because each mineral has a set of unique d-spacings. Typically, this is achieved by comparison of d-spacings with standard reference patterns.All diffraction methods are based on in an X-ray tube.
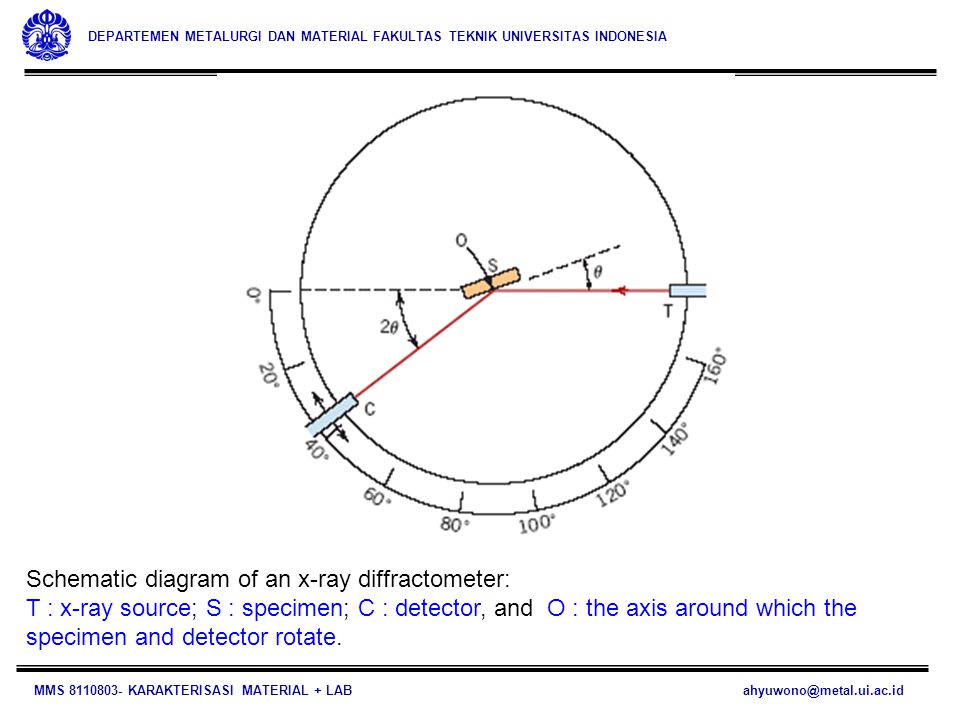
These X-rays are directed at the sample, and the diffracted rays are collected. A key component of all diffraction is the angle between the incident and diffracted rays. Powder and single crystal diffraction vary in instrumentation beyond this. X-ray Powder Diffraction (XRD) Instrumentation - How Does It Work? X-ray powder diffractogram. Peak positions occur where the X-ray beam has been diffracted by the crystal lattice. The unique set of d-spacings derived from this patter can be used to 'fingerprint' the mineral.The geometry of an X-ray diffractometer is such that the sample rotates in the path of the collimated X-ray beam at an angle θ while the X-ray detector is mounted on an arm to collect the diffracted X-rays and rotates at an angle of 2 θ.
The instrument used to maintain the angle and rotate the sample is termed a goniometer. For typical powder patterns, data is collected at 2 θ from 5 ° to 70 °, angles that are preset in the X-ray scan. X-ray powder diffraction is most widely used for the identification of unknown crystalline materials (e.g. Minerals, inorganic compounds). Homogeneous and single phase material is best for identification of an unknown.
Must have access to a standard reference file of inorganic compounds (d-spacings, hkls). Requires tenths of a gram of material which must be ground into a powder. For mixed materials, detection limit is 2% of sample. For unit cell determinations, indexing of patterns for non-isometric crystal systems is complicated. Peak overlay may occur and worsens for high angle 'reflections'User's Guide - Sample Collection and Preparation. Data Collection The intensity of diffracted X-rays is continuously recorded as the sample and detector rotate through their respective angles.
A peak in intensity occurs when the mineral contains lattice planes with d-spacings appropriate to diffract X-rays at that value of θ. Although each peak consists of two separate reflections (K α 1 and K α 2), at small values of 2 θ the peak locations overlap with K α 2 appearing as a hump on the side of K α 1. Greater separation occurs at higher values of θ. Typically these combined peaks are treated as one. The 2 λ position of the diffraction peak is typically measured as the center of the peak at 80% peak height. Data ReductionResults are commonly presented as peak positions at 2 θ and X-ray counts (intensity) in the form of a table or an x-y plot (shown above). Intensity ( I) is either reported as peak height intensity, that intensity above background, or as integrated intensity, the area under the peak.
The relative intensity is recorded as the ratio of the peak intensity to that of the most intense peak ( relative intensity = I/I 1 x 100 ). Determination of an UnknownThe d-spacing of each peak is then obtained by solution of the Bragg equation for the appropriate value of λ. Once all d-spacings have been determined, automated search/match routines compare the ds of the unknown to those of known materials. Because each mineral has a unique set of d-spacings, matching these d-spacings provides an identification of the unknown sample. A systematic procedure is used by ordering the d-spacings in terms of their intensity beginning with the most intense peak. Files of d-spacings for hundreds of thousands of inorganic compounds are available from the as the Powder Diffraction File (PDF). Many other sites contain d-spacings of minerals such as the.
Commonly this information is an integral portion of the software that comes with the instrumentation. Determination of Unit Cell DimensionsFor determination of unit cell parameters, each reflection must be indexed to a specific hkl. LiteratureThe following literature can be used to further explore X-ray Powder Diffraction (XRD). Bish, DL and Post, JE, editors.
Modern Powder Diffraction. Reviews in Mienralogy, v.
Mineralogical Society of America. Cullity, B. Elements of X-ray diffraction. Addison-Wesley, Reading, Mass. Klug, H.
X-ray diffraction procedures for polycrystalline and amorphous materials. Wiley, New York. Moore, D.
Reynolds, Jr. X-Ray diffraction and the identification and analysis of clay minerals.
Oxford University Press, New York.Related LinksFor more information about X-ray Powder Diffraction (XRD) follow the links below.; Materials Research Lab, University of California- Santa Barbara.; an on-line journal that describes and demonstrates a wide range of applications using Xray diffraction.University of California, Santa Barbara.from LLNL.from Steve Nelson, Tulane University.part of the. Use the 'Learn About' link to find animations of the structures of common molecules (including minerals), crystallography learning resources (tutorials, databases and software), resources on crystallization, and tutorials on symmetry and point groups.Teaching Activities and ResourcesTeaching activities, labs, and resources pertaining to X-ray Powder Diffraction (XRD). This problem develops skills in X-ray diffraction analysis as applied to clay mineralogy, reinforces lecture material on the geochemistry of weathering, and demonstrates the role of petrologic characterization in site engineering. (PowerPoint 1.6MB Sep7 07) by Melody Bergeron, at Montana State University. Brady, John B., and Boardman, Shelby J., 1995, Introducing Mineralogy Students to X-ray Diffraction Through Optical Diffraction Experiments Using Lasers.
Education, v. 43 #5, 471-476. Brady, John B., Newton, Robert M., and Boardman, Shelby J., 1995, New Uses for Powder X-ray Diffraction Experiments in the Undergraduate Curriculum. Education, v. 43 #5, 466-470. Dutrow, Barb, 1997, Better Living Through Minerals X-ray Diffraction of Household Products, in: Brady, J., Mogk, D., and Perkins D. (eds.) Teaching Mineralogy, Mineralogical Society of America, p.
349-359. Hovis, Guy, L., 1997, Determination of Chemical Composition, State of Order, Molar Volume, and Density of a Monoclinic Alkali Feldspar Using X-ray Diffraction, in: Brady, J., Mogk, D., and Perkins D. (eds.) Teaching Mineralogy, Mineralogical Society of America, p. 107-118. Brady, John B., 1997, Making Solid Solutions with Alkali Halides (and Breaking Them), in: Brady, J., Mogk, D., and Perkins D. (eds.) Teaching Mineralogy, Mineralogical Society of America, p.
91-95. Perkins, Dexter, III, and Sorensen, Paul, Mineral Synthesis and X-ray Diffraction Experiments, in: Brady, J., Mogk, D., and Perkins D. (eds.) Teaching Mineralogy, Mineralogical Society of America, p. 81-90. Hollecher, Kurt, A Long-Term Mineralogy Practical Exam, in: Brady, J., Mogk, D., and Perkins D. (eds.) Teaching Mineralogy, Mineralogical Society of America, p.
43-46. Hluchy, M.M., 1999, The Value of Teaching X-ray Techniques and Clay Mineralogy to Undergraduates, Jour. Geoscience Education, v.
 RSS Feed
RSS Feed
